The breeding scheme should have two main aims:
- The maintenance of the colony so as to ensure genetic integrity with the minimum amount of genetic change each generation.
- An operationally simple and flexible breeding method with no unnecessary record keeping.
General organisation of the colonies
Where the strain is being bred on a large scale (say over 50 young per week) the colony should be divided up into:
- A self-perpetuating Foundation Stock (FS) colony of 10-50 pairs. Surplus animals go into:
- Up to three generations of Multiplication colonies which do not contribute to the long-term survival of the strain. These can be bred as trios. They can be random mated unless the aim is to identify new mutations as they arise. Three generations of multiplication is usually sufficient to produce large numbers of experimental animals.
Cages can, optionally, be given coloured labels such as white for FS, and green, amber and red to distinguish each generation. All the offspring from red-labelled cages (the third multiplication generation) are used for research
Note that this is in strong contrast to the breeding of outbred stocks which should be maintained as one large colony.
Maintenance of the Foundation Stock (FS) colony
The FS colony should consist of permanently mated monogamous pairs, except in those species (e.g. hamsters, rabbits, chickens) where such matings are not practical either because of fighting or for economic reasons. Each pair should be identified by a number, and should have a paper or electronic record card. Full and detailed records of origins and breeding performance should be kept for each pair in the FS colony as this will be useful in tracking the source of any problems that may arise.
A pedigree chart should also be kept for each colony. The chart is a useful means of keeping the history of the stock, and is particularly useful in selection of future breeding stock within the FS colony. An EXCEL spreadsheet would probably be suitable for keeping such a chart if charting is not part of a comprehensive electronic breeding package.
Selection of breeding stock
Breeding stock should be selected on the basis of the pedigree relationships shown in a pedigree chart and on the average reproductive performance of each subline (a subline is a branch tracing back to a common ancestor) taken as a whole. It is important to select for good breeding performance in order to try to eliminate any new mutations which have adverse effects on general vigor, although it is unlikely that such selection will actually improve performance.
In the hypothetical chart below there are three sub-lines derived from pairs coloured red, blue and green. The mean breeding performance of all animals derived from these three pairs should be calculated, and offspring from the one with the highest mean should be bred from.
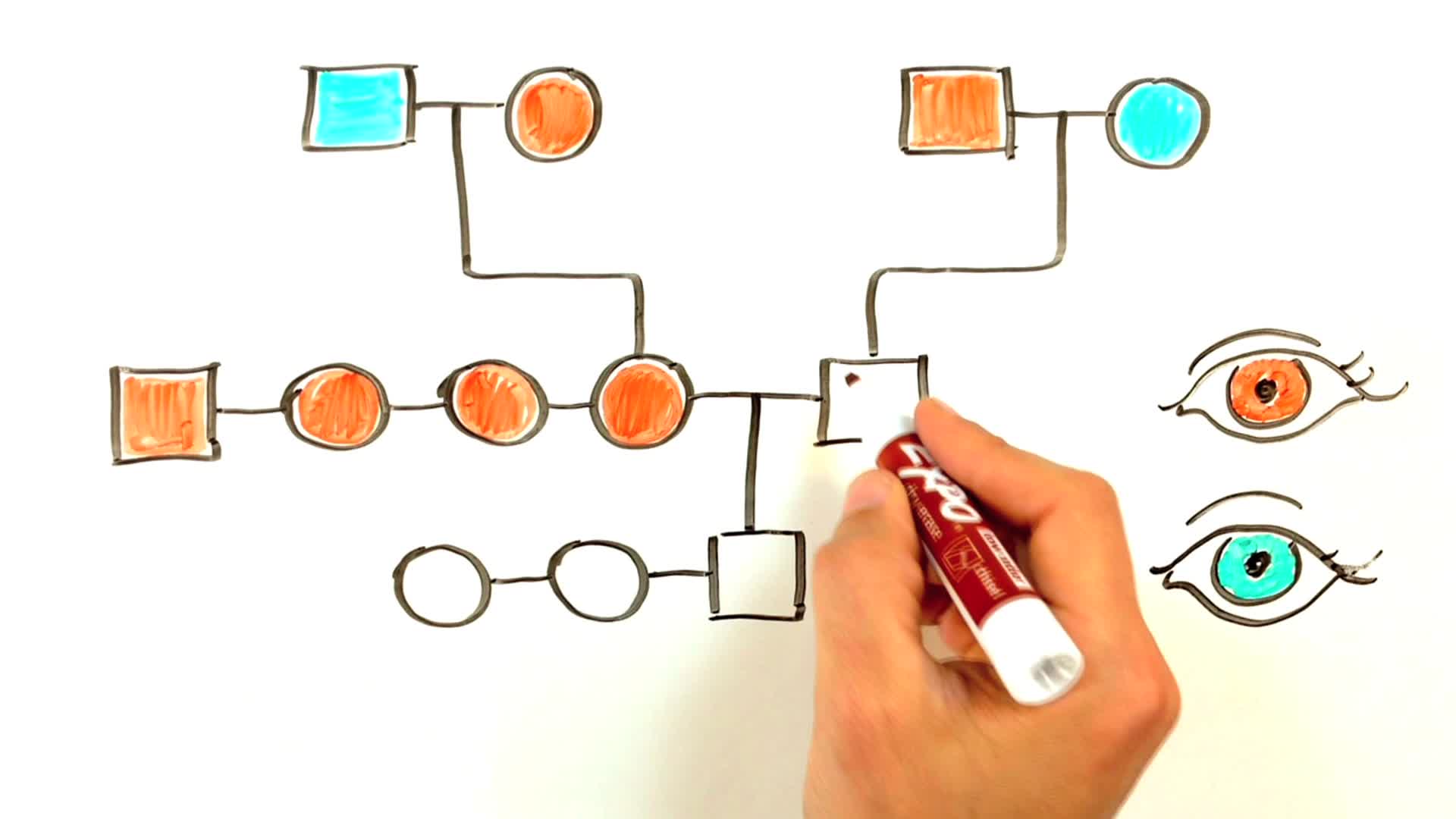 An index of the reproductive performance of every pair at the end of its breeding life or at an intermediate stage where necessary can be recorded on the pedigree chart. The index used could be the total young weaned by the pair per week of breeding life, sometimes also called the “Production Efficiency Index” or PEI.The pedigree chart should be arranged so that all pairs of the current FS generation trace back to a common ancestor 5-7 generations back (NOTE: other authorities recommend that all the current generation should trace back to a common ancestor 3-5 generations back; however, this may result in the inadvertent fixation of unwanted new mutations). This is a very small colony with a maximum of eight pairs per generation. If the sub-line derived from the red pair had the best average breeding performance, then offspring from this branch would need to be multiplied up for a few generations before further between-line was practiced.
An index of the reproductive performance of every pair at the end of its breeding life or at an intermediate stage where necessary can be recorded on the pedigree chart. The index used could be the total young weaned by the pair per week of breeding life, sometimes also called the “Production Efficiency Index” or PEI.The pedigree chart should be arranged so that all pairs of the current FS generation trace back to a common ancestor 5-7 generations back (NOTE: other authorities recommend that all the current generation should trace back to a common ancestor 3-5 generations back; however, this may result in the inadvertent fixation of unwanted new mutations). This is a very small colony with a maximum of eight pairs per generation. If the sub-line derived from the red pair had the best average breeding performance, then offspring from this branch would need to be multiplied up for a few generations before further between-line was practiced.
Thus the PEI (y/f/wk) = (total weaned x 7)/ total days in production. Other indices of productivity may be used if found to be more convenient.
Note that a sudden and substantial increase in breeding performance might indicate genetic contamination and should be treated with considerable suspicion. Similar calculations can be made where records are kept electronically.
The selection of future breeding stock in the FS colony should have absolute priority over the supply of animals to the expansion colony or to provide experimental animals, and control of the FS colony should be by a geneticist, research scientist, animal curator or a trained animal technician. It should not be left to a junior untrained caretaker.
Maintenance of the multiplication colony
The aim of the multiplication colony is to produce a large number of uniform animals as economically as possible. Random mating rather than brother x sister mating may be used on the grounds that new recessive mutants are more likely to be “covered up” in the heterozygous state with random mating, thus leading to greater uniformity.
As exact pedigree relationships are not usually important, trio matings, or some other mating scheme which is more economical of space than brother x sister mating, may be used. However, some institutions wish to find any spontaneous mutations which may occur, and in this case the multiplication colony should be maintained as a “pedigree expansion colony” with monogamous pairs and brother x sister mating.
Usually, the only records needed are those required to control the husbandry and regulate the output of the colony. A simple individual cage record and a weekly summary of the total output of the MC are probably all that is needed.
A “control chart”, also known as a “Shewart chart” gives a good visual display of and trends in production, which might indicate future problems. Control charts typically have a horizontal line drawn at the mean and an upper and lower control limit (UCL and LCL, respectively) drawn at three standard deviations from the mean. Warning limits may also be drawn at two standard deviations from the mean.
The figure below is a control chart of the PEI for a colony of BALB/c mice over a period of one year. The mean was 0.98 young per female per week, but there were quite large fluctuations. However, there was no evidence of a trend. If one point was outside the outer red line, or two consecutive points were outside the inner red bars, then an investigation would be in order.
Charts of this sort are available in some statistical packages such as MINITAB, but can easily be drawn by hand or similar charts could be devised using a spread-sheet.
Careful control of the generation of the MC should be exercised either by physical separation of the first, second, etc., generation colonies or by using coloured cage labels as recommended shown above.